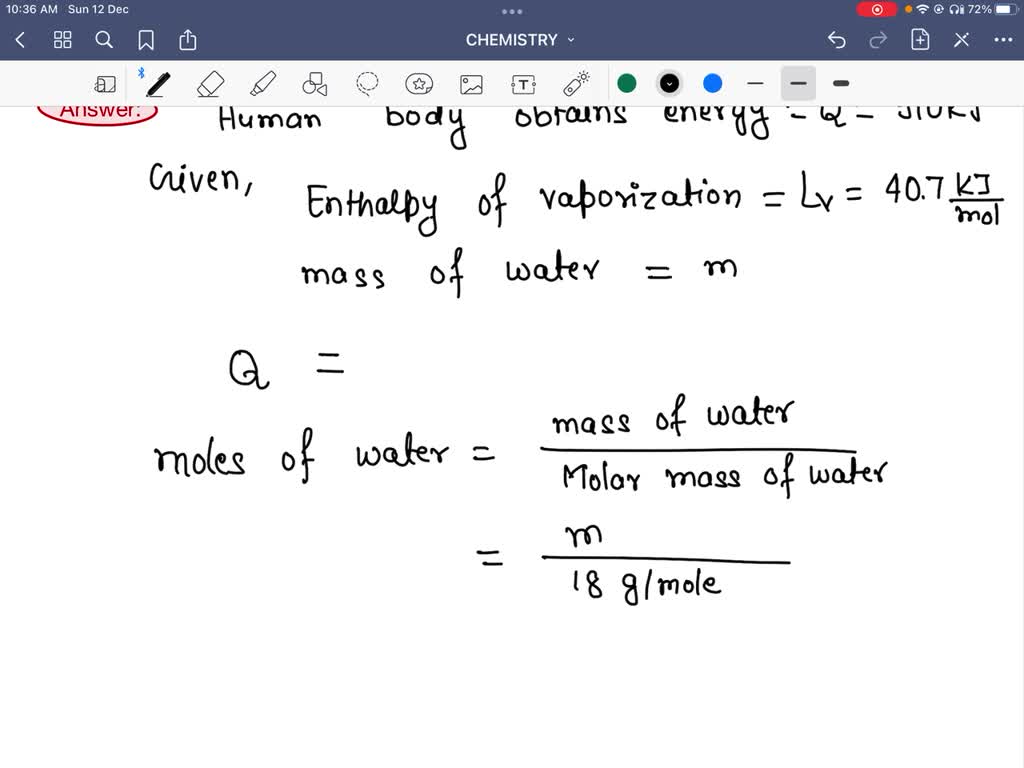
SOLVED: The human body obtains 910 kJ of energy from a candy bar. If this energy were used to vaporize water at 100.0 ?C, how much water (in liters) could be vaporized?

Life-like” stance: a1. infant and a2. adult female (House of the gold... | Download Scientific Diagram

Vaping health effects and the human anatomy breathing vaporized steam from an electronic cigarette concept in a 3D illustration style Stock Photo - Alamy

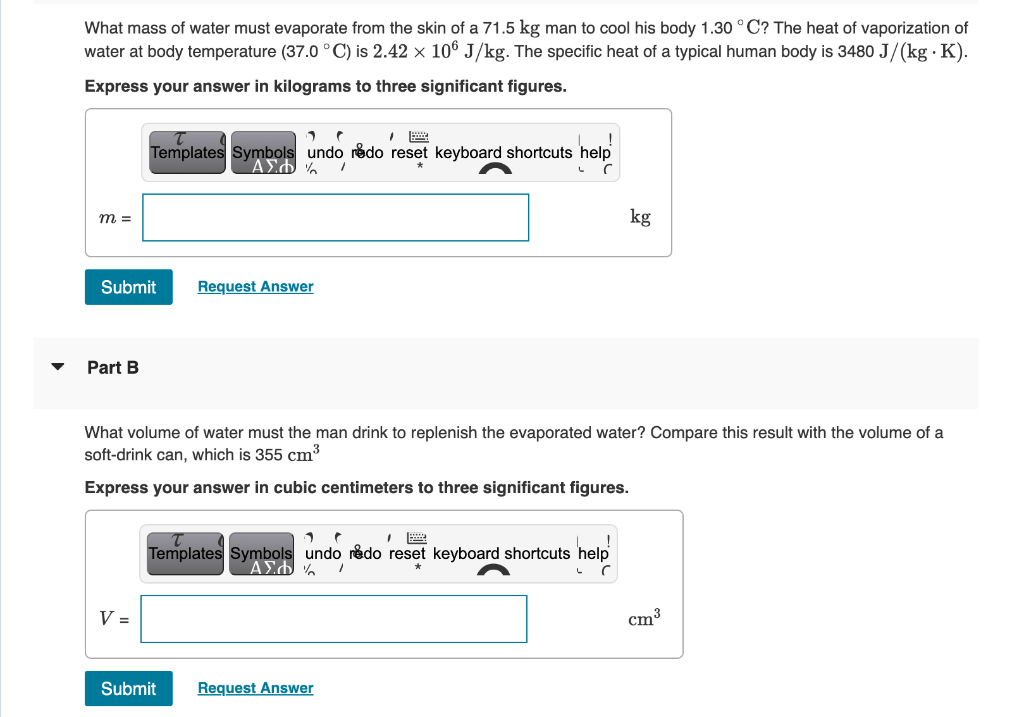
SOLVED: When perspiration on the human body absorbs heat, some of the perspiration turns into water vapor. The latent heat of vaporization at body temperature (37^∘C) is 2.42 × 10^6 J /

Schematic representation of the human body, with the intake of food,... | Download Scientific Diagram
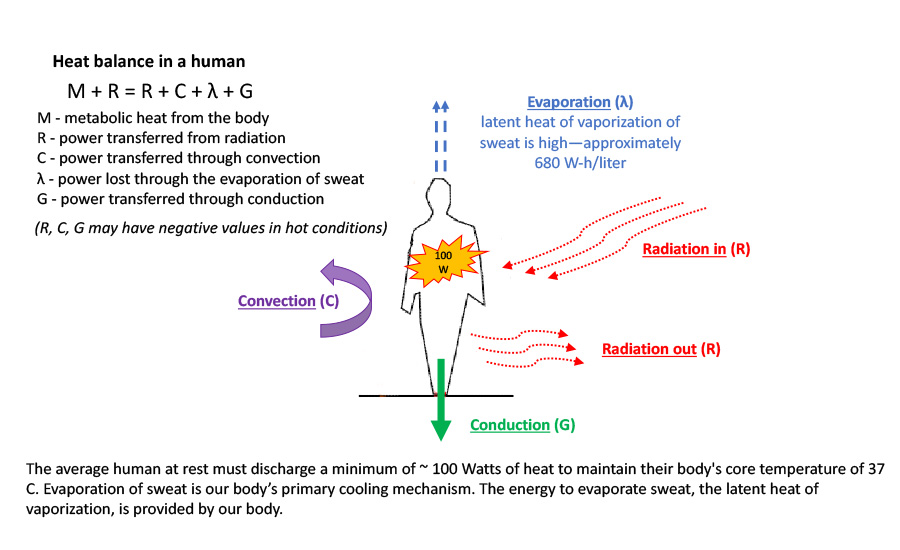
We Can Learn a Lot from Our Bodies When it Comes to Efficiently Cooling Buildings | 2018-09-06 | Engineered Systems Magazine