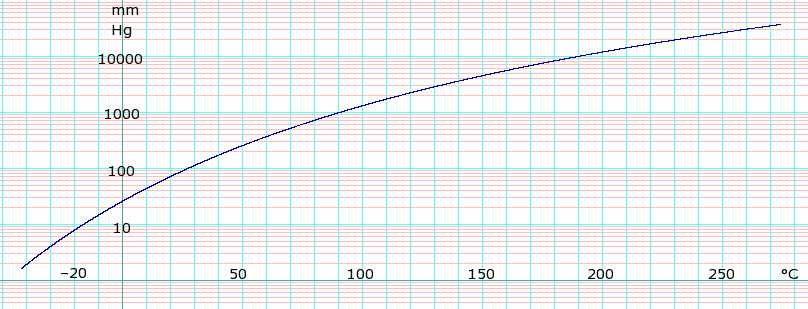
Vapor pressure measurement and correlation of acetonitrile + 1-butyl-3-methylimidazolium chloride, + 1-butyl-3-methylimidazolium tetrafluoroborate, and + 1-hexyl-3-methylimidazolium chloride - ScienceDirect

Table 2 from Acetonitrile ( CH 3 CN ) infrared absorption cross sections in the 3 m m region | Semantic Scholar
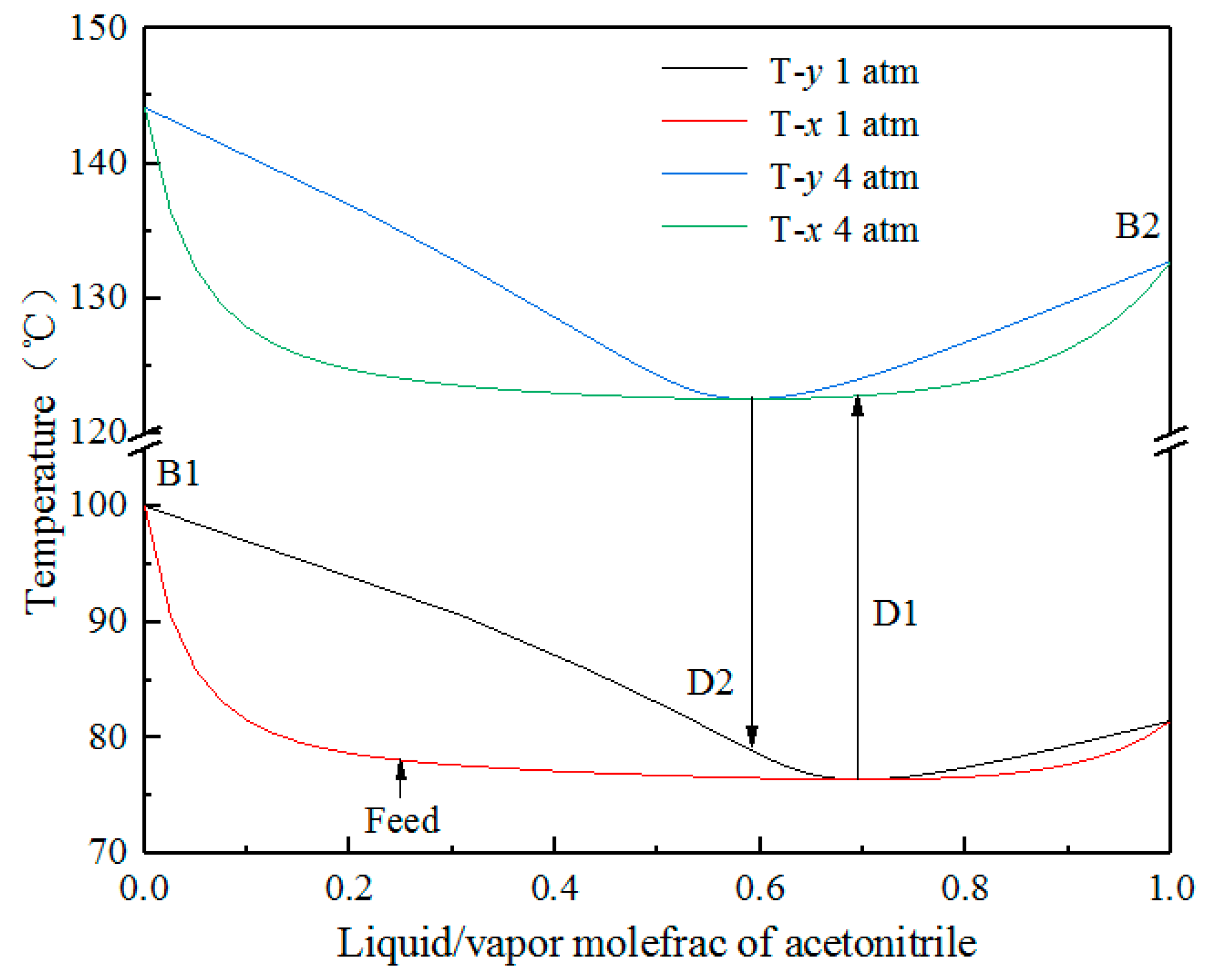
Processes | Free Full-Text | Process Simulation of the Separation of Aqueous Acetonitrile Solution by Pressure Swing Distillation

Processes | Free Full-Text | Vapor–Liquid Equilibrium in Binary and Ternary Azeotropic Solutions Acetonitrile-Ethanol-Water with the Addition of Amino Esters of Boric Acid

SOLVED: The vapor pressures of acetone (1) and acetonitrile (2) can be evaluated by the Antoine equations: In P1 = 14.5463 - 35.93/T In P2 = 14.2724 - 49.15/T where T is

Measurement and correlation of isobaric vapour-liquid equilibrium for the ( acetonitrile + water) system containing different ionic liquids at atmospheric pressure - ScienceDirect
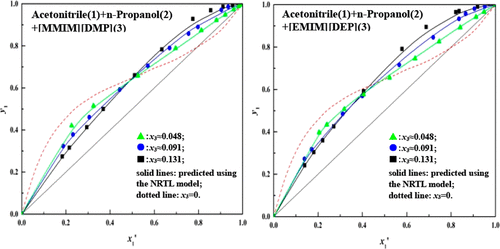
Isobaric Vapor–Liquid Equilibrium of the Acetonitrile + 1-Propanol + Ionic Liquids at an Atmospheric Pressure,Journal of Chemical & Engineering Data - X-MOL

SOLVED: At 23.0°C, the vapor pressure of acetonitrile, CH3CN, is 180.0 torr while that of acetone, C3H6O, is 85.0 torr. What is the vapor pressure of a solution which contains 0.550 moles

Vapor pressure of acetonitrile for various temperatures. Simulation... | Download Scientific Diagram